Confused about COVID-19 boosters? Experts answer 6 common questions
By Michael Merschel, American Heart Association News
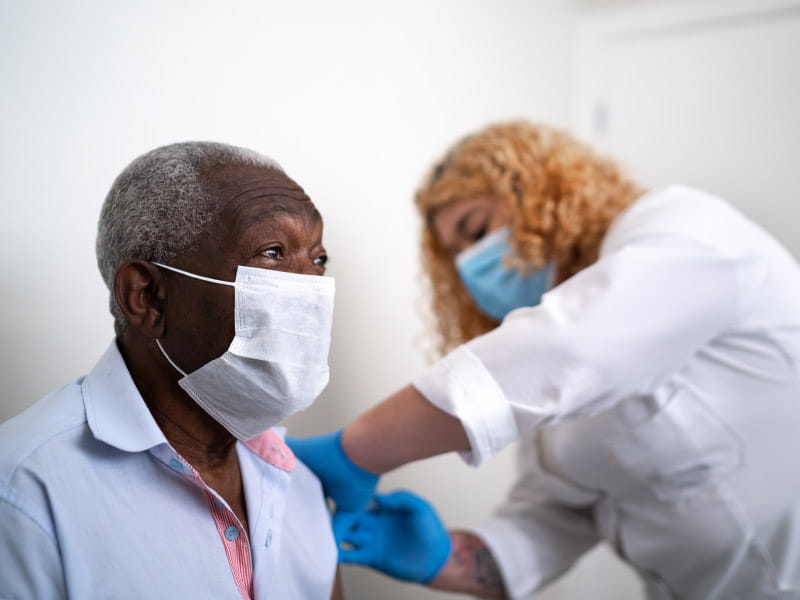
When the Food and Drug Administration recently authorized a second COVID-19 booster shot for some people, many of those eligible wondered whether to get one – and when, given that cases are once again rising in some parts of the country.
Here's are answers to six common questions to clear up some of the confusion.
Who is eligible for a second booster?
Based on the FDA's decision, the Centers for Disease Control and Prevention now says:
– People age 12 and up who are immunocompromised may choose to receive a second booster dose of an mRNA COVID-19 vaccine (Moderna or Pfizer) at least four months after the first booster dose.
– People 50 and older may choose to have a second booster if it has been at least four months since their first booster.
– People 18 and up who received the Johnson & Johnson vaccine and booster may get an mRNA booster at least four months after their first booster.
The CDC already recommended a primary series of COVID-19 vaccination for everyone age 5 and older, and a booster for everyone 12 and older. But the optional tone of the language around the second booster has left many people wondering what to do, health care experts say.
Do I need a second booster?
Dr. Won Lee, medical director of Boston Medical Center's Home Care Program, provides home care to about 500 people in their 80s and above. She tells them, "Yes, you need it."
Although a study published last month in the New England Journal of Medicine suggests a fourth vaccine dose provided only marginal benefits in a group of healthy, young health care workers, pharmacist Orly Vardeny said older people in general benefit from boosters because as people age, their immune response to vaccination weakens.
Anyone with chronic health conditions "is certainly on that list" of people who need a fresh booster, said Vardeny, an associate professor of medicine at the Minneapolis VA Health Care System and University of Minnesota. That would include people with cardiovascular disease.
But she said even for people 50 and older with no underlying conditions, "there's really not a good reason not to get boosted" again.
Why did the booster recommendations change?
The changes are based on new science, Vardeny said. "I think if they didn't change things on us, despite a changing landscape of COVID, that would be worrisome."
The CDC still says vaccines provide important protection against serious illness, hospitalization and dying from COVID-19, especially among those who received a booster dose.
Should I delay a second booster until I want to travel, or until another surge in my area?
A study published April 5 in the New England Journal of Medicine showed that while a second booster in people 60 and older protected against severe illness during a six-week period, protection against infection began to wane after four weeks.
Still, Vardeny, who has done extensive research on flu vaccines, doesn't recommend trying to outthink the coronavirus.
"If we've learned anything about COVID-19, it's that it's unpredictable," she said. "And so, while we may think we can predict the next surge, we truly don't know when it will occur. And I think if it occurs, we want our immune system to be in the best shape it can possibly be."
Lee said if you're 50 or older, in good health, live in an area with a low rate of COVID-19, "and you just want to wait a little bit, I don't think that's unreasonable."
But she doesn't encourage delaying. Trying to align the timing of a vaccine with variables such as vaccine availability and a person's individual immunity is "kind of gambling a little bit," she said. "It seems a little unnecessary. So I would just go ahead and get it."
Is it safe to mix and match vaccines?
For people who initially received the Pfizer or Moderna vaccine, switching to the other brand for a booster is safe, but Lee said there's not enough information to make a strong recommendation. "I think there is some early data to say that it might be helpful, but it probably isn't harmful either."
From a logistical perspective, Vardeny said, people should get whatever is available at their pharmacy.
But she is in favor of the CDC's advice that people who received the Johnson & Johnson vaccine should switch to an mRNA vaccine for their second booster.
Are the boosters ever going to end?
COVID-19 isn't going away, Vardeny said. "I think people should start getting mentally prepared for the fact that this may become endemic, similar to influenza." And the new normal is likely to include boosters. The FDA could start making decisions on the next phase of boosters as early as May.
Lee, who said too many people still need their first booster or even initial vaccinations, hopes people think of others when making booster decisions.
"My plea is always to focus on the more vulnerable folks in the community," she said. Even people who feel strong and healthy, or who already had COVID-19, should be mindful about risks to children younger than 5 who can't be vaccinated yet and to older adults, she said. "Vaccinating everyone helps, especially those who are less able to protect themselves."
Editor's note: Because of the rapidly evolving events surrounding the coronavirus, the facts and advice presented in this story may have changed since publication. Visit Heart.org for the latest coverage, and check with the Centers for Disease Control and Prevention and local health officials for the most recent guidance.
If you have questions or comments about this American Heart Association News story, please email [email protected].
 Login
Login





